Research Project 1:
A role for human WIPI3 in lysosomal phosphoinositide signaling
Principal Investigator
Prof. Dr. rer. nat. Tassula Proikas-Cezanne
PhD Student (FOR2625 funded)
PhD Student (associated)

Project Summary
Lysosomal function and autophagy critically contribute to secure the maintenance of cellular homeostasis and many human diseases are characterized by autophagy/lysosome dysfunction. Inaccurate phosphoinositide signaling, in particular pathways involving phosphatidylinositol 3-phosphate (PI3P) and phosphatidylinositol 3,5-bisphosphate (PI(3,5)P2), impact both autophagy and lysosomal function. In order to contribute to the understanding of the roles of PI3P and PI(3,5)P2 in the control of autophagy and lysosome function we addressed the function of the human WIPI (WD-repeat protein interacting with phosphoinositide) proteins (WIPI1-WIPI4), PI3P- and PI(3,5)P2-binding proteins that we identified earlier, in the process of autophagy. We showed that all four WIPI proteins fulfill distinct scaffold functions in the signal control of autophagy and at the nascent autophagosome. Whereas WIPI1 and WIPI2 function as PI3P effectors at the nascent autophagosome, WIPI3 and WIPI4 function in the signal control of autophagy upstream of PtdIns3P production. In response to AMPK activation, WIPI4 is released from a WIPI4-ATG2/AMPK-ULK1 complex and translocates, together with ATG2, to the nascent autophagosome for size controlling membrane expansion, to which WIPI3 also contributes. Upstream and on lysosomes, WIPI3 associates the TSC complex, which controls lysosomal TORC1 activity (Bakula et al., Nat Commun. 2017). Here we will address the role of human WIPI3 as PI3P/PI(3,5)P2 effector in lysosomal signaling.
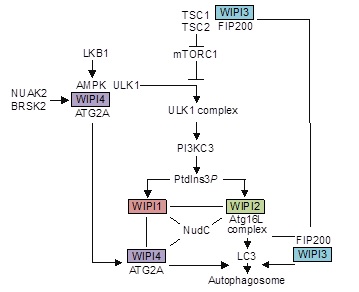
Working model (modified from Bakula et al., Nat Commun 2017)
References
Bakula D, Müller AJ, Zuleger T, Takacs Z, Franz-Wachtel M, Thost AK, Brigger D, Tschan MP, Frickey T, Robenek H, Macek B, Proikas-Cezanne T. WIPI3 and WIPI4 β-propellers are scaffolds for LKB1-AMPK-TSC signalling circuits in the control of autophagy. Nat Commun 2017; 8:15637.
Proikas-Cezanne T, Takacs Z, Dönnes P, Kohlbacher O. WIPI proteins: essential PtdIns3P effectors at the nascent autophagosome. J Cell Sci 2015; 128:207-17.
Proikas-Cezanne T, Waddell S, Gaugel A, Frickey T, Lupas A, Nordheim A. WIPI-1alpha (WIPI49), a member of the novel 7-bladed WIPI protein family, is aberrantly expressed in human cancer and is linked to starvation-induced autophagy. Oncogene 2004; 23:9314-25.
